Product Description
MSI FFPE Reference Set is a reference material for microsatellite instability (MSI) loci. These biomarkers are tumor-specific, show a high frequency in colorectal cancer and are stable across different ethnicities.
MSI screening is used to see if the Lynch syndrome genes are working properly. Microsatellites are regions of repeated DNA that change in length (show instability) when mismatch repair is not working properly. MSI testing looks at the length of certain DNA microsatellites from the tumor sample to see if they have gotten longer or shorter as a measure of instability (1).
MSI definition from the National Cancer Institute: A change that occurs in certain cells (such as cancer cells) in which the number of repeated DNA bases in a microsatellite (a short, repeated sequence of DNA) is different from what it was when the microsatellite was inherited. Microsatellite instability may be caused by mistakes that don’t get corrected when DNA is copied in a cell. It is found most often in colorectal cancer, gastric cancer, and endometrial cancer, but it may also be found in many other types of cancer (2).
Product Overview
Features:
- 100% based on cell lines (no plasmids or artificial sequences added)
- MSI cell content quantitated via ddPCR
- Supportive in MSI assay validation and routine quality control for several systems
- Manufactured under ISO13485 condititions
Intended use:
MSI FFPE Reference Set is intended as commutable, qualitative control material for microsatellite instability (MSI) markers in FFPE format based on human cell lines. It consists of MSI FFPE MSI-H control with all MSI markers and MSI FFPE MSS control with 0% frequency for all MSI markers. This Reference set enables monitoring of the performance, procedures and workflow of laboratory tests using FFPE in combination with the extraction workflows.
The product is intended to be used only with an MSI Test System by professionals.
Loci covered:
- Biocartis Panel: ACVR2A, BTBD7, DIDO1, MRE11, RYR3, SEC31A, and SULF2
- NCI Panel: BAT25, BAT26, MONO-27, NR-21, NR-24
- Promega Panel (including NCI Panel): BAT25, BAT26, MONO-27, NR-21, Penta C, Penta D, NR-24
Format: FFPE (Formalin Fixed Paraffin Embedded)
1 set contains 2 products:
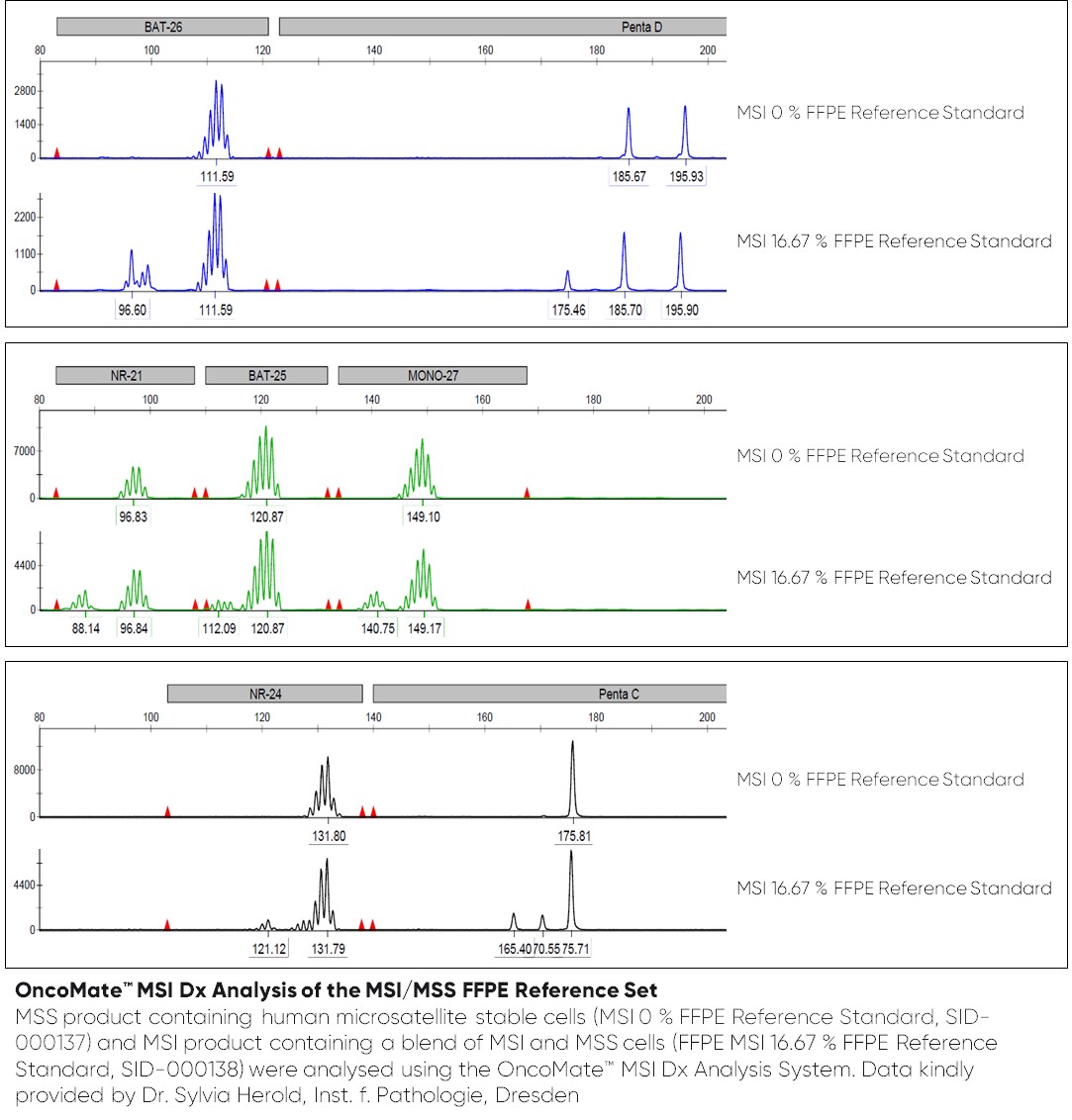
- SID-000137, MSI 0 % FFPE Reference Standard
- SID-000138, MSI 16.67 % FFPE Reference Standard
Unit size: 1 curl per vial (2 curls in total per set)
Expected DNA yield: ≥ 110 ng (using Qiagen QIAamp DNA FFPE Tissue Kit)
DNA Integrity Number (DIN): ≥ 5
Thickness of one curl/cut: 10 µm
Storage: 2-8 °C
Expiry: stable for 24 months from date of manufacturing (as supplied)
1: information provided by the CDC
2: information provided by the NIH